Ace Tips About Does Polarity Mean Charge

Unraveling Polarity and Charge
1. Understanding the Basics
So, you're pondering the relationship between polarity and charge? It's a great question! Many people use these terms somewhat interchangeably, which can lead to confusion. Let's clear things up with a dash of science and a sprinkle of plain English. Think of it this way: electricity is like the human body, and charge and polarity are two of its essential organs. Charge is what powers everything, whereas polarity is how the body uses the power.
At its heart, "charge" refers to a fundamental physical property that causes matter to experience a force when placed in an electromagnetic field. We typically talk about two types of charge: positive and negative. It's this imbalance of positive and negative charges that leads to the fascinating world of electricity and magnetism. So, charge is the fundamental "stuff" that interacts.
Imagine you're building with LEGOs. Charge is like the individual LEGO bricks themselves. You can have different colors and sizes, but they're all still LEGO bricks. Similarly, you can have positive and negative charges, but they're both still "charge."
Now, where does "polarity" come in? That's where things get interesting. Polarity describes the distribution of charge. It's about having distinctly positive and negative regions in a system, like a battery with a positive (+) end and a negative (-) end. Or, think about a water molecule. It has a slight negative charge near the oxygen atom and a slight positive charge near the hydrogen atoms.
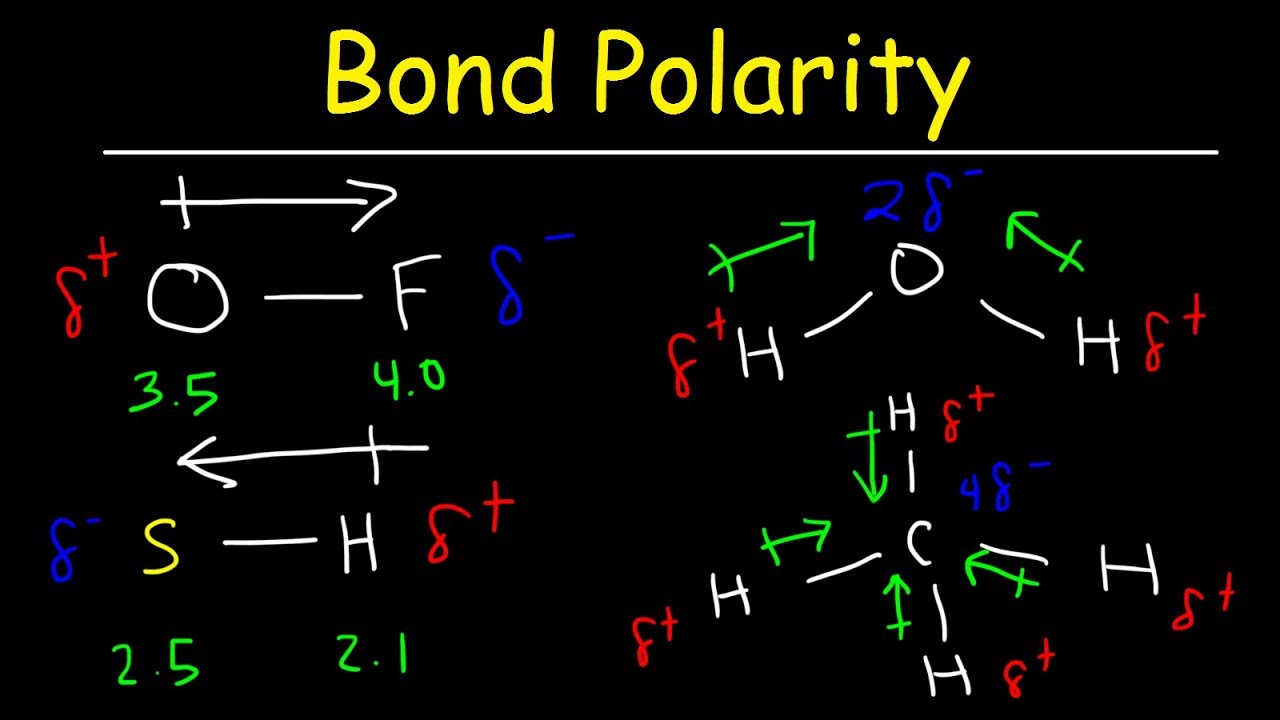
Polarity Of A Molecule Explained
Polarity
2. The Uneven Distribution of Electron Density
Polarity isn't just about having opposite ends; it's about an uneven distribution of electron density. Electrons, being negatively charged, are drawn to regions where they can achieve a more stable configuration. This unevenness creates a dipole moment — a measure of the separation of positive and negative charges. A molecule is considered polar if it has a net dipole moment.
Think of it like a tug-of-war. If both sides are pulling with equal force, the rope stays in the middle. But if one side is significantly stronger, the rope moves towards them. In a polar molecule, some atoms pull on the electrons more strongly than others, creating that uneven distribution.
Take water, for example (H2O). Oxygen is more electronegative than hydrogen, meaning it has a stronger pull on electrons. This causes the oxygen atom to have a slight negative charge and the hydrogen atoms to have slight positive charges, making water a polar molecule. This polarity is responsible for many of water's unique properties, like its ability to dissolve many substances.
This difference in electron density also explains why some things mix and others dont. Ever try mixing oil and water? Because water is polar and oil is nonpolar (meaning it has an even distribution of charge), they dont want to hang out together. Polar things like to mix with polar things, and nonpolar things with nonpolar things. Its like a social gathering for molecules!

Charge is Fundamental, Polarity is Derived
3. The Subtle but Significant Distinction
The key takeaway is that charge is a fundamental property, while polarity is a derived property. You can't have polarity without charge, but you can have charge without polarity. A single proton, for instance, has a positive charge, but it doesn't have polarity because there isn't a separation of positive and negative regions within itself.
Imagine you have a box full of positively charged particles. The box, as a whole, has a net positive charge. However, unless the particles are arranged in a specific way to create distinct positive and negative regions, the box doesn't exhibit polarity. It's just a box full of positive charge.
To further illustrate, consider an ionic crystal like sodium chloride (NaCl). The sodium ions (Na+) have a positive charge, and the chloride ions (Cl-) have a negative charge. While there are charged particles present, the crystal as a whole might not exhibit a strong polarity unless there's an external electric field influencing the distribution of those charges. The individual ions have charge, but the macroscopic object requires some distribution difference to exhibit polarity.
So, while charge is the "ingredient," polarity is the "recipe" that determines how that ingredient is used to create something with distinct positive and negative characteristics. Thinking about them in this way can help to avoid some serious head-scratching later on!

Why Does This Matter?
4. The Real-World Impact of Polarity
Understanding the difference between charge and polarity is crucial in many areas of science and technology. In chemistry, it helps us understand how molecules interact with each other, predicting solubility, reactivity, and physical properties. In electronics, it's fundamental to understanding how circuits work, how batteries function, and how semiconductors behave.
For example, the polarity of solvents is critical in chemical reactions. If you're trying to dissolve a polar compound, you'll need a polar solvent. Likewise, if you're working with a nonpolar compound, you'll need a nonpolar solvent. The "like dissolves like" rule is all about polarity!
In biology, polarity plays a vital role in the structure and function of biological molecules like proteins and DNA. The polarity of amino acids, the building blocks of proteins, determines how proteins fold and interact with other molecules. The polarity of DNA influences its structure and how it interacts with enzymes during replication and transcription.
Even in everyday life, polarity is all around us. From the batteries that power our devices to the detergents we use to clean our clothes, polarity plays a role in how things work. So, understanding the difference between charge and polarity isn't just an academic exercise; it's essential for understanding the world around us!

Polarity Definition & Examples Britannica
In Short
5. A Quick Recap
Let's wrap this up with a quick recap. Charge is the fundamental property that causes matter to experience a force in an electromagnetic field. It comes in two types: positive and negative. Polarity, on the other hand, describes the distribution of charge, specifically the uneven distribution that creates distinct positive and negative regions. Charge is fundamental, and polarity is derived.
Remember the LEGO analogy? Charge is the LEGO bricks, and polarity is how you arrange those bricks to create something with distinct features. Or the tug-of-war analogy? Charge is the people pulling the rope, and polarity is the uneven pulling that moves the rope to one side.
While the terms are often used loosely, understanding the distinction between charge and polarity is essential for understanding many scientific and technological phenomena. So, the next time you hear someone talk about charge or polarity, you'll know exactly what they're talking about!
Hopefully, this has cleared up the confusion between charge and polarity. Now, go forth and impress your friends with your newfound knowledge! Just don't be too smug about it. After all, learning is a lifelong journey, and there's always more to discover.

FAQ
6. Answering Your Burning Questions
Okay, let's tackle some frequently asked questions to further solidify your understanding.
Q: Can something have a charge but no polarity?
A: Absolutely! A single ion, like a sodium ion (Na+), has a charge but doesn't have polarity. Polarity requires a separation of positive and negative regions within a system.
Q: Is polarity the same as being ionic?
A: Not quite. Ionic compounds are formed through the transfer of electrons, resulting in charged ions. These ions create strong electrostatic attractions, leading to the formation of an ionic bond. While ionic compounds exhibit charge separation, polarity refers more broadly to the uneven distribution of electron density in a molecule, which can occur even in covalent bonds.
Q: How does polarity affect the boiling point of a liquid?
A: Polar molecules tend to have higher boiling points than nonpolar molecules of similar size and mass. This is because the positive and negative regions of polar molecules attract each other, creating stronger intermolecular forces that require more energy to overcome and transition into the gaseous phase.
Q: What are some examples of polar and nonpolar molecules?
A: Water (H2O) is a classic example of a polar molecule, as is ammonia (NH3). Nonpolar molecules include methane (CH4) and oil, which have an even distribution of electron density.