Fine Beautiful Info About Why Does IE Increase Left To Right
Understanding The Periodic Table Through Lens Of Volatile Group
Unlocking the Mystery
1. What's the Buzz About Ionization Energy, Anyway?
Ever wondered why certain elements are more likely to hog electrons or give them away? A big part of the answer lies in something called ionization energy (IE). Think of it like this: ionization energy is the amount of "oomph" — the energy — needed to yank an electron away from an atom in its gaseous state. Yes, gaseous state is very important because we don't want any outside interference with our ionization process.
The higher the ionization energy, the harder it is to remove that electron. It's like trying to steal candy from a very grumpy baby dragon. Some dragons just won't let go of their precious sweets, while others are more willing to share (or, you know, just have them taken).
Now, why do we care? Well, ionization energy is a key indicator of how reactive an element will be. Elements with low ionization energies tend to lose electrons easily and form positive ions (cations), while those with high ionization energies hold onto their electrons tightly and might be more inclined to gain electrons and form negative ions (anions). Knowing this helps us predict how elements will interact with each other, forming compounds and generally making the world go 'round.
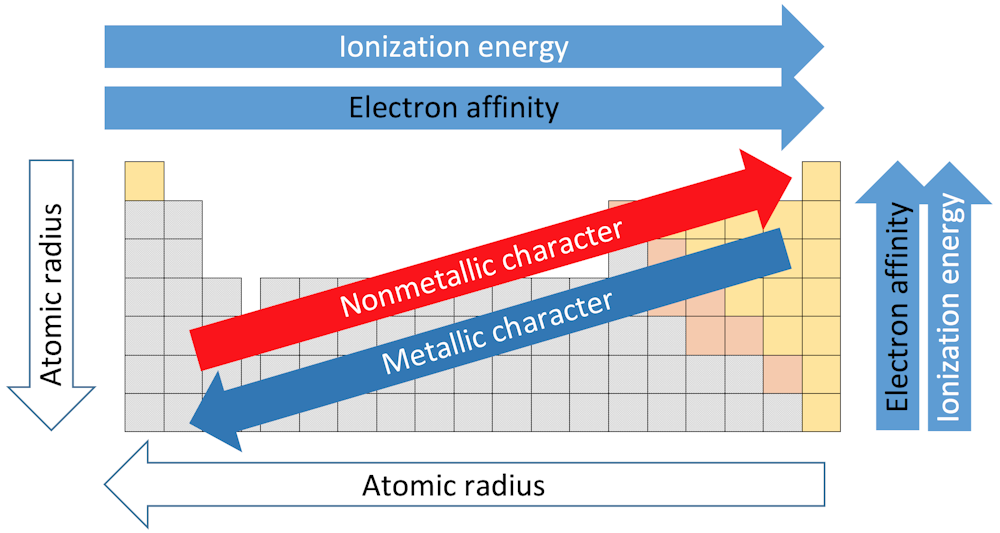
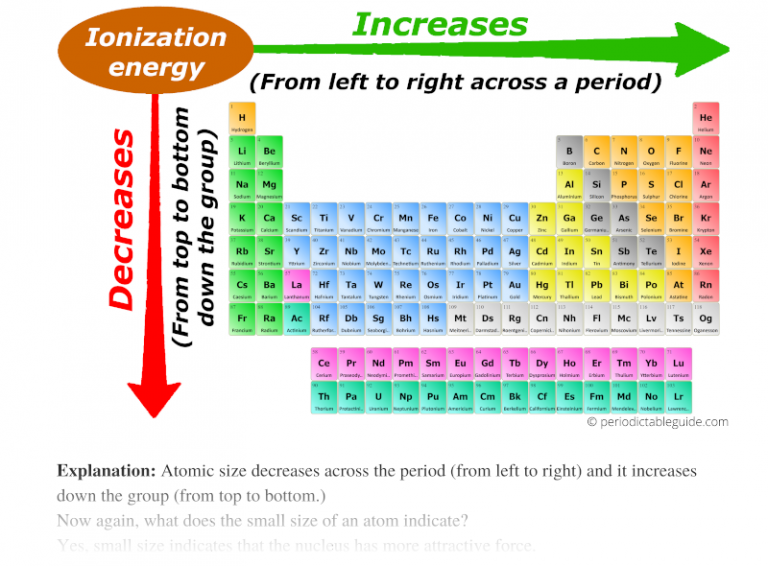
So, where does left and right fit in the ionization energy? On the periodic table, there's a clear trend. As we move from left to right across a period, ionization energy generally increases. Let's unravel the reasons behind this trend, because, like most things in chemistry, there are always exceptions to the rule!

Imperfect But Chosen Part 1 Real Families, Faith Pastor Mark
The Core Culprits
2. Digging Deeper
At the heart of this trend is the concept of "effective nuclear charge." Imagine the nucleus of an atom as a super-powerful magnet pulling on all the electrons. As we move across a period (left to right), the number of protons in the nucleus increases. This means the positive charge of the nucleus gets stronger and stronger. That's what we call increase left to right.
But wait, there's more! The number of electrons also increases, but here's the kicker: they're all being added to the same energy level or "shell." Since theyre in the same shell, the increased nuclear charge has a greater hold on each individual electron, pulling them closer to the nucleus. Its like adding more weight to one side of a tug-of-war; the electrons get pulled in more forcefully.
This increased attraction makes it harder to remove an electron, hence the higher ionization energy. Think of it as trying to pull a magnet away from an even stronger magnet. It's going to take a lot more effort, right?
Effective nuclear charge, in essence, is the net positive charge experienced by an electron in a multi-electron atom. It's the actual "pull" an electron feels after accounting for the shielding effect, which we'll tackle next.
3. The Shielding Effect
So, the nucleus is pulling, but not all electrons feel the full force of that pull. That's because inner-shell electrons act as a shield, partially blocking the outer electrons from experiencing the full nuclear charge. This is called the "shielding effect."
As we move across a period, the number of core electrons (those inner-shell electrons) remains roughly the same. This means the shielding effect doesn't change significantly. Therefore, the increased nuclear charge has a more pronounced effect on the valence electrons (the outermost electrons), leading to a stronger attraction and higher ionization energy.
Think of it like this: imagine you're trying to see a stage performance, but there's a row of people in front of you. Those people are shielding you from the stage. If the number of people in front of you stays the same, but the spotlight on the stage gets brighter (like the increasing nuclear charge), it's going to be easier for you to see the performance, even with the people in front.
In simpler terms, the combination of increased nuclear charge and relatively constant shielding makes it increasingly difficult to remove electrons as you move from left to right across the periodic table. It's like trying to peel an onion with increasingly stronger glue holding the layers together.
Easy Defiintions For Words Associated With The Periodic Table Bowers
Exceptions to the Rule
4. The Case of Group 15 and 16 Elements
Ah, the exceptions! Just when you think you understand a trend, chemistry throws you a curveball. One notable exception occurs between Groups 15 (Nitrogen group) and 16 (Oxygen group) elements. Nitrogen, phosphorus, arsenic, etc. (Group 15) have slightly higher ionization energies than their counterparts in Group 16, even though Group 16 comes "later" in the period.
Why the hiccup? It's all about electron configuration. Group 15 elements have a half-filled p subshell. This half-filled configuration is particularly stable because each p orbital contains only one electron. Because of Hund's rule that like electron to each get their own p-orbital. Removing an electron from this stable configuration requires more energy than removing one from Group 16 elements, which already have one p orbital that's paired.
Imagine you're organizing socks. A half-filled drawer of individual socks is pretty neat and tidy. Trying to take one out is a bit disruptive. But a drawer with some pairs and some singles is already a little messy. Taking a single sock out isn't such a big deal.
Essentially, the extra stability of the half-filled p subshell in Group 15 outweighs the effect of the slightly higher nuclear charge in Group 16, resulting in the observed "dip" in ionization energy.
5. The Group 2 and 13 Elements
A similar, although less pronounced, blip occurs between Group 2 (alkaline earth metals) and Group 13 (boron group) elements. Group 2 elements often have slightly higher ionization energies than expected based solely on the left-to-right trend.
In this instance, we're looking at a filled s subshell versus starting a new p subshell. Group 2 elements have a completely filled s subshell (s2). This filled subshell provides extra stability. It requires more energy to remove one of these s electrons than it does to remove the first p electron from the Group 13 elements.
Again, consider the sock drawer. A drawer full of neatly paired socks is quite satisfying. Yanking one of those pairs apart takes some effort. But if you just started a new, mostly empty drawer, adding a single sock (the p electron) doesn't require as much disruption.
These exceptions remind us that while trends are useful, we always need to consider the specific electronic configurations of the elements involved. Chemistry loves to keep us on our toes!

So, What Does It All Mean? The Significance of IE Trends
6. Predicting Chemical Behavior
Understanding ionization energy trends isn't just an academic exercise; it has real-world implications for predicting how elements will behave chemically. As we've seen, elements with low ionization energies tend to lose electrons easily, becoming positive ions and readily forming ionic compounds with elements that readily gain electrons (high electron affinity).
For example, alkali metals (Group 1) have very low ionization energies and readily react with halogens (Group 17), which have high electron affinities, to form salts like sodium chloride (NaCl). This is a direct consequence of their differing abilities to lose and gain electrons, driven by their respective ionization energies and electron affinities.
Conversely, elements with high ionization energies are less likely to lose electrons and may be more prone to forming covalent bonds, where electrons are shared rather than transferred. Think of noble gases (Group 18), which have extremely high ionization energies and are notoriously unreactive. They simply don't want to lose any of their precious electrons!
By knowing the ionization energy of an element, we can make informed predictions about its reactivity, the types of bonds it will form, and the compounds it will create. It's like having a cheat sheet to the chemical world!
7. Beyond Basic Chemistry
The applications of ionization energy extend beyond simple chemical reactions. It's used in various analytical techniques, such as mass spectrometry, to identify and quantify different elements and compounds. In mass spectrometry, molecules are ionized, and then their mass-to-charge ratio is measured. The ionization energy plays a crucial role in this process.
Ionization energy also finds its way into materials science, where it helps in understanding the electronic structure of materials and predicting their properties. For example, the ionization energy of a semiconductor material can affect its ability to conduct electricity. Knowledge of ionization energy is even used in astrophysics to understand the composition and conditions of stars and other celestial objects.
From predicting the behavior of elements in a test tube to analyzing the composition of distant stars, ionization energy is a powerful tool with wide-ranging applications. It's a testament to the fundamental principles that govern the behavior of matter and the universe around us.
In conclusion, the general increase in ionization energy from left to right across the periodic table is due primarily to the increase in effective nuclear charge, coupled with a relatively constant shielding effect. However, exceptions exist and should be understood, because there are always exceptions!
Frontiers Native Valve, Prosthetic And Cardiac Devicerelated
Frequently Asked Questions (FAQs)
8. Q1
A1: Yes, generally ionization energy increases as you move up a group (column) on the periodic table. This is because the valence electrons are closer to the nucleus, experiencing a stronger attraction, and there's less shielding from inner electrons. The atom gets smaller as you go up the group, leading to a tighter hold on the outer electrons.
9. Q2
A2: Understanding ionization energy helps in many areas! It's used to predict chemical reactivity, design new materials with specific electronic properties (like semiconductors), and even analyze the composition of stars. Mass spectrometry, a common analytical technique, relies heavily on ionization energy principles.
10. Q3
A3: Yes! The first ionization energy (IE1) is always the lowest because it's the energy required to remove the first electron from a neutral atom. Subsequent ionization energies (IE2, IE3, etc.) involve removing electrons from positively charged ions, which requires more energy due to the increased attraction between the nucleus and the remaining electrons. So, IE1 < IE2 < IE3, and so on.
